

Reshoring is a competitive advantage.
Evolve Manufacturing helps medical device teams bring production closer—reducing risk, improving responsiveness, and maintaining control in an increasingly volatile environment.
→ 75,000 sq. ft. facility with 3,000 sq. ft. of cleanrooms
→ Faster response times: local, collaborative manufacturing
→ Reduced supply chain risk and improved execution
ISO 13485:2016 Certified • FDA Registered Facility
Why bring manufacturing closer?
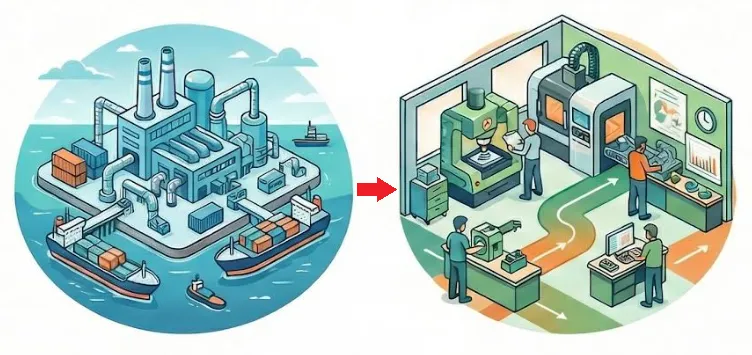
In today’s environment, the risks of offshore manufacturing continue to increase—impacting quality, lead times, and overall execution.
For products that are still evolving, distance slows decision-making and introduces unnecessary complexity. Local partners = more control.
Reshoring isn’t just a sourcing decision—it’s an operational advantage.


Why Choose Evolve Manufacturing?

• Reduced inventory by millions and significantly lowered monthly overhead for OEM partners
• Proven ability to scale production rapidly while maintaining consistent on-time delivery and quality standards
• Delivered high-reliability manufacturing with zero defects across extended production periods
• Accelerated production readiness from initial setup to full output in weeks, not months
What do we support?

Medical Device Manufacturing – Complex assemblies, regulated builds, and production support across device categories
Biotech & Life Sciences Equipment – Capital equipment and instrument builds requiring precision and consistency
Sub-Assemblies & Systems Integration – High-quality assembly and integration for critical components


How Does The Process Work?

Step 1: Initial Discussion
Step 2: Program Review
Step 3: Build & Support